Michael Mozer BS1
Guy Raz, MD2
Ryan Wyatt, MD2
Alexander Toledo, DO, PharmD2
1University of New England College of Osteopathic Medicine
Biddeford, ME USA
2Department of Emergency Medicine
Maricopa Medical Center, Phoenix, AZ USA
Abstract
Hypothermia can progress quickly and become life threatening if left untreated. Rewarming in the severely hypothermic patient is of critical importance and is achieved with active and passive techniques. Here we present a case of a hypothermic patient with cardiac instability for whom thoracic lavage was ultimately used. We will review the treatment of hypothermia and discuss the technical aspects our approach.
Case Presentation
A 53 year-old male with a past medical history of substance abuse, chronic hepatitis C, and poorly controlled type 2 diabetes mellitus complicated by a recent hospitalization for osteomyelitis was brought to the emergency department after being found lying on a road in a shallow pool of water in the early morning hours of a rainy day in Phoenix, Arizona. The ambient temperature that night was 39 °F (3.9 °C). Emergency Medical Services (EMS) noted a decreased level of consciousness and obtained a finger stick glucose of 15 mg/dl. EMS reported a tympanic membrane temperature of 23.9 °C. En route, the patient was administered 2mg naloxone and 25g dextrose intravenously with no improvement in his mental status. On Emergency Department (ED) arrival, the patient had a GCS of 8 (Eyes 4, Verbal 1, Motor 3) and exhibited intermittent posturing. His foot wound appeared clean and without signs of infection. The initial core temperature recorded was 25.9°C via bladder thermometer, systolic blood pressure was 92/50, and heart rate fluctuated between 50 and 90 beats per minute.
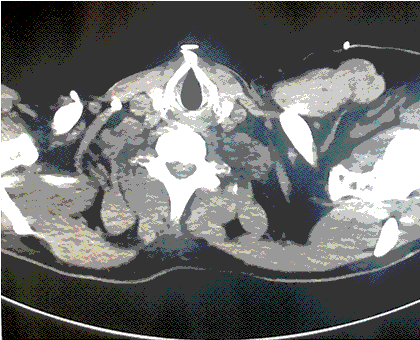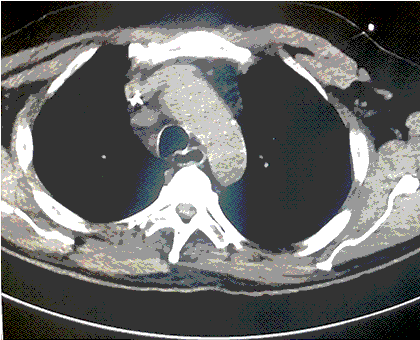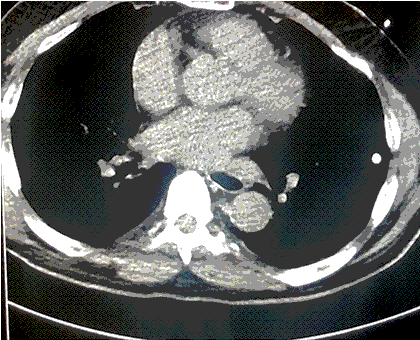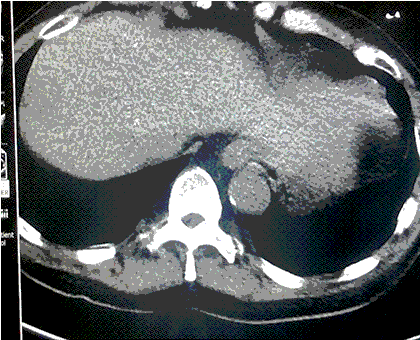
After removing wet clothing, initiation of warmed saline, and placing a forced warm air blanket on the patient, he was intubated for airway protection and vasopressors were initiated. Osborn waves were evident on the initial EKG (Figure 1).

Figure 1. Initial EKG with Osborn Waves (arrows).
A warmed ventilator circuit was initiated with only 0.5 °C increase in temperature in first 30 minutes. Despite these measures, he remained hypotensive and unstable. Significant laboratory findings were a white blood cell count of 25.5 thousand (92% neutrophils), lactic acid of 7.6, potassium of 5.8, serum creatinine of 1.05, glucose of 283, INR of 1.1, and urine drug screen positive for cocaine. Given his recalcitrance to norepinephrine and risk of death secondary to fatal dysrhythmia with temperatures below 28 °C intrathoracic lavage initiated.
The right hemithorax was selected for irrigation because left-sided tube placement can induce ventricular fibrillation in a perfusing patient (1). Using standard sterile technique, two 36 French thoracostomy tubes were placed; the first in the second intercostal space along the mid-clavicular line, and the second in the 5th intercostal space in the posterior axillary line (1-3). The tips of the thoracostomy tubes were oriented such that the anterior-superior tube was positioned near the right apex and the lateral-inferior tip was positioned low in the thoracic cavity (1,3). To maintain the temperature of the instilled fluid, a fluid warmer system (Level 1; Smiths Medical; Minneapolis, MN) was used and set to 41 °C. A Christmas tree adapter was used to connect the IV tubing to the superior thoracostomy tube, and a water seal chamber was attached to the inferior tube for passive drainage (3). Flow through the system was targeted to maintain steady passive drainage as described in the literature (1-6).
Thoracic cavity lavage with 41 °C saline was performed and the patient was transferred to the medical ICU after 3 hours in the ED. When he was transferred his core temperature was 29 °C and he remained on norepinephrine for hemodynamic instability. After 2 hours of continued rewarming in the MICU, his core temperature was 32 °C. Osborn waves evident on initial EKG were resolved (Figure 2).

Figure 2. Repeat EKG showing resolution of Osborn waves.
The patient left against medical advice from the hospital 4 days later neurologically intact and without sequela.
Discussion
Hypothermia can be clinically classified as mild, moderate or severe (7). Mild hypothermia, defined as core temperatures of 32-35 °C, presents with shivering. Amnesia, dysarthria, ataxia, tachycardia, and tachypnea can also be seen (1). Moderate hypothermia, defined as core temperatures of 28-32 °C, usually can present with or without shivering. Stupor, hypoventilation, paradoxical undressing and non-fatal arrhythmias such as atrial fibrillation and junctional bradycardia may also be seen (1). Patients with severe hypothermia, generally defined as temperatures below 28 °C, can present with coma, areflexia, pulmonary edema, bradycardia, and hypotension (1). There is a significant risk of fatal cardiac dysrhythmias without rapid therapeutic rewarming (1,7,8).
Rewarming in the hypothermic patient is of critical importance and is achieved with passive and/or active techniques. Attempts to limit heat loss are often unsuccessful, especially in the absence of a normal shiver response. It however remains as the first line treatment for hypothermia (8-10). Passive rewarming is attempted by the removal of cold/wet clothing and keeping the patient covered (8-10). Active external rewarming (AER) is the next line of treatment and consist of the use of externally rewarming devices such as warmed blankets, warm environment, forced air warming (Bair Hugger; 3M; Maplewood, MN) or warm hot water bladders placed in the groin and axilla (1,7-10). Active Internal Rewarming (AIR) techniques can be used to achieve more rapid increases in core temperature and are primarily utilized in cases of cardiac instability or if AER is unsuccessful (8). When available, the method of choice for active internal rewarming (AIR) is cardiopulmonary bypass (CPB) or extracorporeal membrane oxygenation (ECMO) as they can achieve the fastest increase in core temperature (9 °C/hr and 6 °C/hr respectively) and provide cardiovascular support (1,8,11,12). Several techniques are described in the literature that can be considered if CPB or ECMO are unavailable. These include esophageal warming devices, endovascular catheters, hemodialysis, and endocavitary lavage (1,2,4-6,13-15). While no randomized controlled trials exist, several case reports and reviews have tried to compare various techniques. These sources to do not seem to favor any particular technique over another but rather reports the rates of temperature rise (1-3,5-7,13-15). Classically, lavage techniques are attempted in the thoracic cavity, the peritoneum, the bladder, the stomach, the esophagus, or the colon. These techniques are generally coupled with warm IV fluids and warming air through the ventilator to limit loss of body heat to iatrogenic procedures during the rewarming attempt (1,7). Thoracic lavage is effective with a reported rewarming rates of 3-6 °C/hr and with excellent outcomes in case reports (1,2,4-6). Here we present a case of a hypothermic patient with cardiac instability where thoracic lavage is used and discuss the technical aspects of this approach.
Our case demonstrates the efficacy of utilizing thoracic cavity lavage for rapid rewarming in patients with severe hypothermia with a pulse and at high risk of fatal cardiac arrhythmia. In multiple case reports, thoracic lavage has been used successfully in hypothermic patients who suffered complete cardiopulmonary collapse requiring CPR (2,4,5). Although warm thoracic lavage is not the treatment of choice in these circumstances, in a facility not equipped with ECMO or CPB and a patient too unstable to transfer, it seemed to us to be the best technique. Gastric, colonic, and bladder lavage offer very minimal heat transfer due to limitations in surface area (2).
Hemodialysis would have required for us to call in a technician and would have required approval by a nephrologist at our institution. Available central venous rewarming catheters require bypass of a failsafe mechanism that does not allow rewarming to be initiated below 30 °C (1). Peritoneal lavage was a plausible choice but does not directly warm the mediastinum (2). While an open mediastinal technique has been used, we did not feel it was appropriate in a patient with a concurrent pulse (1,3). Thoracic lavage is therefore an effective alternative that should be used in cases where CPB and ECMO are unavailable especially in a patient that is hemodynamically unstable and may not survive transfer. The equipment is readily available to any Emergency Medicine or Critical Care physician. In addition, this case exemplifies the positive outcomes that are associated with rapid rewarming in the hypothermic patient with a pulse. We believe our case demonstrates the efficacy of this technique for myocardial protection from hemodynamic collapse, a topic which has not been adequately studied in the literature.
References
- Brown DJ, Danzl DF. Accidental hypothermia. In: Auerbach PS, ed. Wilderness Medicine. 7th ed. St. Louis: Mosby Inc.; 2017:135-62.
- Plaisier BR. Thoracic lavage in accidental hypothermia with cardiac arrest--report of a case and review of the literature. Resuscitation. 2005 ;66(1):99-104. [CrossRef] [PubMed]
- Schiebout JD. Hypothermic Patient Management. In: Reichman EF. eds. Reichman's Emergency Medicine Procedures, 3e New York, NY: McGraw-Hill. Available at: http://accessemergencymedicine.mhmedical.com/content.aspx?bookid=2498§ionid=201303754 (accessed August 02, 2019).
- Little G. Accidental hypothermic cardiac arrest and rapid mediastinal warming with pleural lavage: A survivor after 3.5 hours of manual CPR. BMJ Case Reports. July 2017:bcr-2017-220900. [CrossRef] [PubMed]
- Turtiainen J, Halonen J, Syväoja S, Hakala T. Rewarming a patient with accidental hypothermia and cardiac arrest using thoracic lavage. Ann Thorac Surg. 2014 Jun;97(6):2165-6. [CrossRef] [PubMed]
- Ellis MM, Welch RD. Severe hypothermia and cardiac arrest successfully treated without external mechanical circulatory support. Am J Emerg Med. 2016;34(9):1913.e5-6. [CrossRef] [PubMed]
- Tintinalli J, Stapczynski J, Ma O, Yealy D, Meckler G, Cline D. Tintinalli's Emergency Medicine. 8th ed. New York, NY: McGraw-Hill Education; 2016:1743-4.
- Brugger H, Boyd J, Paal P. Accidental Hypothermia. N Engl J Med. 2012;367(20):1930-8. [CrossRef] [PubMed]
- Paal P, Gordon L, Strapazzon G, et al. Accidental hypothermia-an update: The content of this review is endorsed by the International Commission for Mountain Emergency Medicine (ICAR MEDCOM). Scand J Trauma Resusc Emerg Med. 2016;24(1):111. [CrossRef] [PubMed]
- Zafren K, Giesbrecht GG, Danzl DF, et al. Wilderness Medical Society practice guidelines for the out-of-hospital evaluation and treatment of accidental hypothermia: 2014 update. Wilderness Environ Med. 2014 Dec;25(4 Suppl):S66-85. [CrossRef] [PubMed]
- Schober A, Sterz F, Handler C, et al. Cardiac arrest due to accidental hypothermia-A 20 year review of a rare condition in an urban area. Resuscitation. 2014;85(6):749-56. [CrossRef] [PubMed]
- Saczkowski RS, Brown DJA, Abu-Laban RB, Fradet G, Schulze CJ, Kuzak ND. Prediction and risk stratification of survival in accidental hypothermia requiring extracorporeal life support: An individual patient data meta-analysis. Resuscitation. 2018;127:51-7.[CrossRef] [PubMed]
- Primozic KK, Svensek F, Markota A, Sinkovic A. Rewarming after severe accidental hypothermia using the esophageal heat transfer device: a case report. Ther Hypothermia Temp Manag. 2018 Mar;8(1):62-4. [CrossRef] [PubMed]
- Murakami T, Yoshida T, Kurokochi A, et al. Accidental hypothermia treated by hemodialysis in the acute phase: three case reports and a review of the literature. Intern Med. 2019 Jun 7. [CrossRef]
- Klein LR, Huelster J, Adil U, et al. Endovascular rewarming in the emergency department for moderate to severe accidental hypothermia. Am J Emerg Med. 2017 Nov;35(11):1624-9. [CrossRef] [PubMed]
Cite as: Mozer M, Raz G, Wyatt R, Toledo A. Severe accidental hypothermia in Phoenix? Active rewarming using thoracic lavage. Southwest J Pulm Crit Care. 2019;19(2):79-83. doi: https://doi.org/10.13175/swjpcc038-19 PDF
 Wednesday, January 1, 2020 at 8:00AM
Wednesday, January 1, 2020 at 8:00AM